Translate this page into:
Pituitary hypointensity: Hemochromatosis as an overlooked cause of hypogonadotropic hypogonadism
*Corresponding author: Rajiv Gupta, Department of Radiodiagnosis, Medanta-The Medicity, Gurugram, Haryana, India. drrajivgupta2014@yahoo.in
-
Received: ,
Accepted: ,
How to cite this article: Garg A, Gupta R, Mothsera M, Modi J. Pituitary hypointensity: Hemochromatosis as an overlooked cause of hypogonadotropic hypogonadism. Case Rep Clin Radiol. doi: 10.25259/CRCR_112_2024
Abstract
Hemochromatosis (HFE) is caused by abnormal accumulation of iron in the parenchymal cells, eventually leading to dysfunction of the involved organs. It can be either primary (or genetic) or secondary (or acquired). Due to the presence of the blood-brain barrier (BBB), iron deposition in the central nervous system is rare but structures lying outside the BBB such as choroid plexus, pituitary, and circumventricular organs are commonly involved. This case series describes 2 cases of pituitary HFE, both presenting with hypogonadotropic hypogonadism, which were diagnosed by magnetic resonance imaging (MRI). In both the cases, T2-weighted and susceptibility-weighted imaging (SWI) images demonstrated markedly low signal in anterior pituitary and choroid plexus with patchy areas of non-enhancement seen on post contrast images. Hence, contrast-enhanced MRI with SWI sequences should be used as non-invasive tool in patients suspected of pituitary gland iron overload.
Keywords
Pituitary hemochromatosis
Hypogonadotropic hypogonadism
Iron deposition
Magnetic resonance imaging
INTRODUCTION
Hemochromatosis (HFE) is caused by abnormal accumulation of iron in the parenchymal cells, eventually leading to dysfunction of the involved organs.[1] The liver, pancreas, heart, skin, and pituitary are some of the most common sites of iron deposition.[2-4] The presence of the blood-brain barrier (BBB) does not allow deposition of excess iron in the central nervous system (CNS), but structures lying outside the BBB such as choroid plexus, pituitary, and circumventricular organs are commonly involved.[5-9] Although, involvement of cortical surface and basal ganglia has also been reported.[5] Iron deposition in various organs leads to liver cirrhosis, diabetes mellitus, bronzing of the skin, cardiomyopathy, and hypogonadism.[1-4,10] We report two cases of pituitary HFE with involvement of choroid plexus, pituitary, basal ganglia, and dentate nuclei of cerebellum in the first case and involvement of choroid plexus and pituitary in second case.
CASE SERIES
Case 1
A 36-year-old male presented with complaints of decreased hair on beard and moustache. No associated comorbidities were noted. On blood examination, ferritin levels were raised to 1260.00 ng/mL (Normal for males: 17.9–464 ng/mL) with reduced total iron binding capacity of 150 ug/dL (Normal range: 265–497 ug/dL). The hormonal workup of the patient was normal. Contrast-enhanced magnetic resonance imaging (MRI) dynamic pituitary was done on Siemens MAGNETOM Verio 3T MRI machine. Anterior lobe of the pituitary gland appeared small in size for age of the patient. The gland appeared intermediate signal intensity on T1-weighted (T1W) and hypointense on T2-weighted (T2W) images [Figure 1a-b]. On dynamic post contrast images, there was normal enhancement of pituitary stalk with patchy areas of non-enhancement seen within the anterior pituitary gland [Figure 1c-d]. Posterior pituitary T1 hyperintense spot was seen at its expected location.
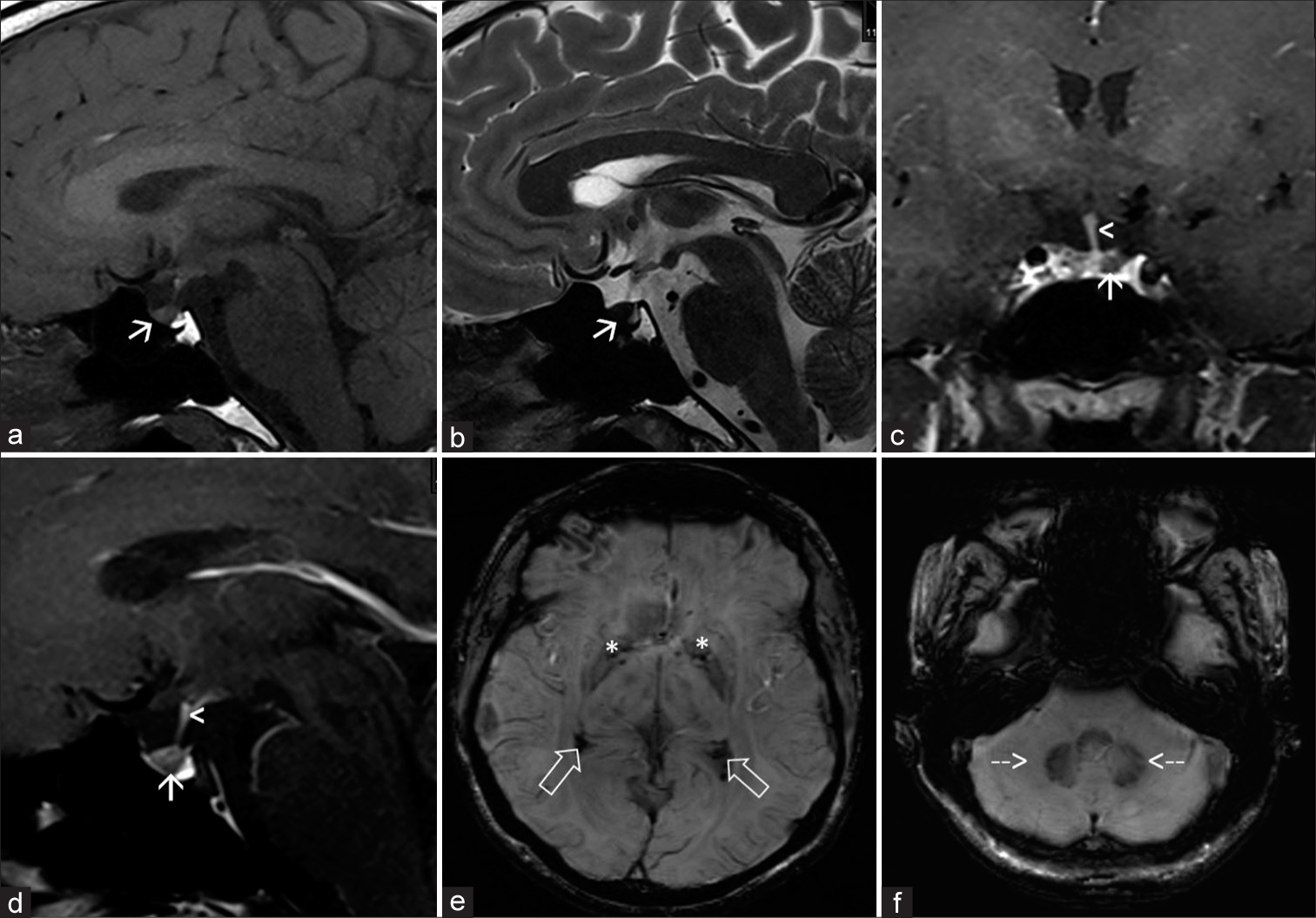
- A 36-year-old male presented with features of hypogonadotropic hypogonadism. On contrast-enhanced magnetic resonance imaging dynamic pituitary. (a) T1 sagittal images showing small intermediate signal intensity anterior pituitary (white arrow) with normal hyperintense spot of posterior pituitary seen. (b) T2 sagittal images showing hypointense anterior pituitary (white arrow). (c and d) T1 coronal and sagittal post contrast images showing patchy areas of non-enhancement within anterior pituitary (white arrows) with normal enhancement of pituitary stalk (white arrowheads) seen. (e and f) Susceptibility-weighted imaging images showing blooming foci in bilateral globus pallidi (white asterisks), trigone of bilateral lateral ventricles (white open arrows), and bilateral dentate nuclei of cerebellum (white dashed arrows).
Bilateral symmetric T2 hypointense and T1 hyperintense signal in the globus pallidi of basal ganglia and the dentate nuclei of the cerebellum was noted. Few tiny blooming foci were seen in the globus pallidi of basal ganglia, bilateral temporal horns and trigones of lateral ventricle, and the dentate nuclei of the cerebellum [Figure 1e-f]. The findings raised suspicion of pituitary iron/hemosiderin deposition. Further, clinical laboratory correlation was suggested. Genetic workup confirmed mutation in HFE gene confirming the diagnosis of primary (genetic) HFE.
Case 2
An 18-year-old female, known case of thalassemia major, on repeated blood transfusions since childhood, presented with complaints of amenorrhea. On blood examination, ferritin levels were raised to 4500.00 ng/mL (Normal for females <50 years: 6.24–137 ng/mL) with reduced total iron binding capacity of 167 ug/dL (Normal range: 265–497 ug/dL). The hormonal workup of the patient was normal. Contrast-enhanced MRI dynamic pituitary with conventional MRI brain sequences was done on Siemens MAGNETOM Aera 1.5T MRI machine. The gland appeared intermediate signal intensity on T1W and hypointense on T2W images [Figure 2a-b]. On post-contrast images, there was normal enhancement of pituitary stalk with lack of enhancement seen within the anterior pituitary gland, [Figure 2c-d] suggesting changes related to pituitary HFE. Blooming seen along the choroid plexus of bilateral lateral ventricles [Figure 2e], third, fourth ventricle, along the foramen of Luschka and Magendie [Figure 2f] was seen likely suggesting iron deposition in clinical settings of thalassemia.
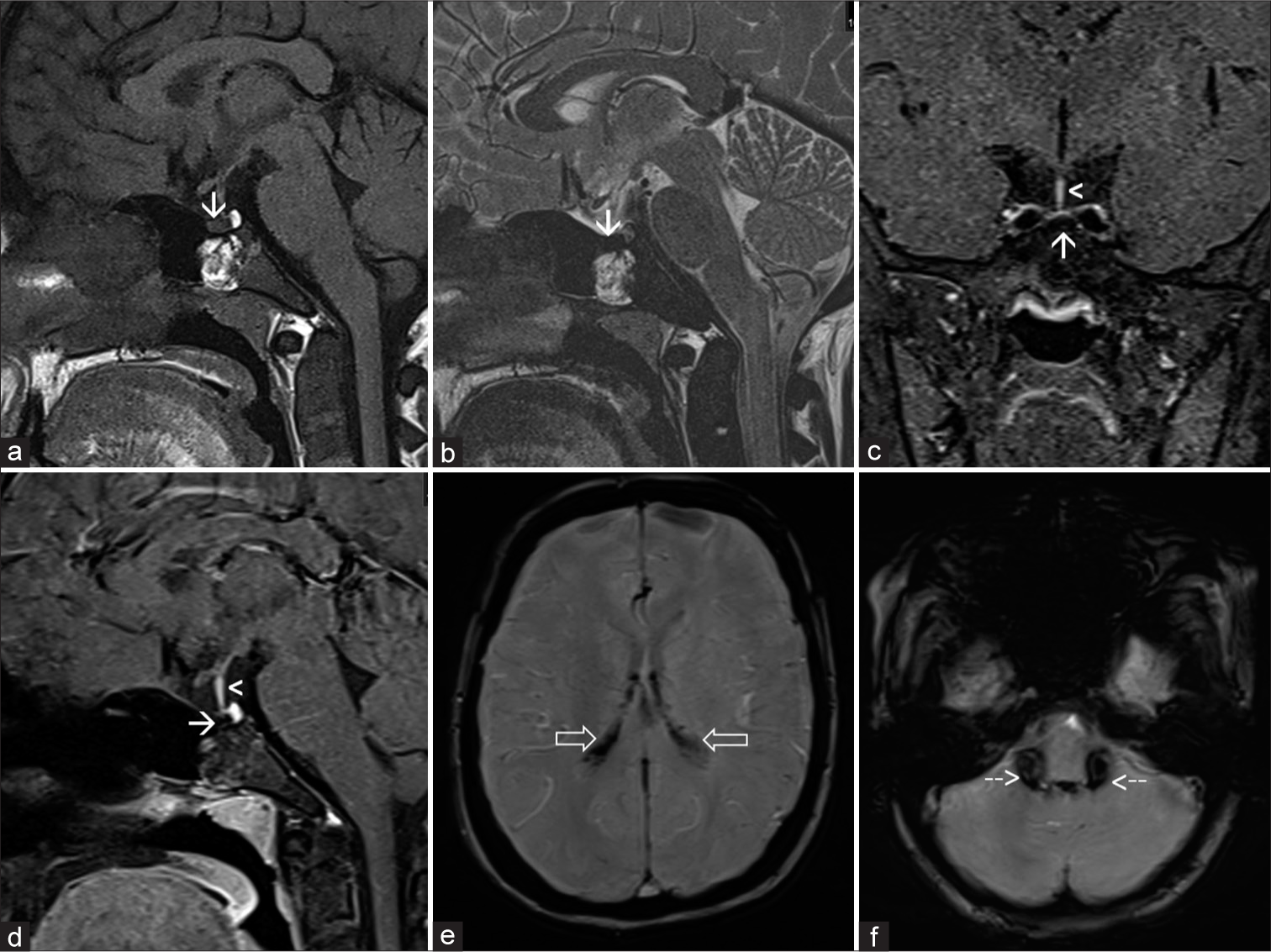
- A 18-year-old female, known case of thalassemia major, on repeated blood transfusions since childhood, presented with complaints of amenorrhea. On contrast-enhanced magnetic resonance imaging dynamic pituitary. (a) T1 sagittal images showing small intermediate signal intensity anterior pituitary (white arrow) with normal hyperintense spot of posterior pituitary seen. (b) T2 sagittal images showing hypointense anterior pituitary (white arrow). (c and d) T1 coronal and sagittal post contrast images showing non-enhancement of anterior pituitary (white arrows) with normal enhancement of pituitary stalk (white arrowheads) seen. (e and f) Susceptibility-weighted imaging images showing blooming foci in bilateral lateral ventricles (white open arrows) and along the foremen of Luschka and Megendie (white dashed arrows).
DISCUSSION
HFE is a systemic disorder causing abnormal accumulation of iron in various organs of the body eventually leading to organ dysfunction. It can be either primary (or genetic) or secondary (or acquired). Primary (or genetic) HFE is most commonly caused due to mutation in HFE gene.[4] Secondary HFE is an acquired cause of iron accumulation most commonly associated with chronic disorders of erythropoiesis, treated with iron and blood transfusions.[1]
Iron plays an important role in cellular function and various physiological processes, such as the production of hemoglobin, oxygen transport, and adenosine triphosphate (ATP) generation. Humans have no physiologic pathway for iron excretion; hence, iron hemostasis is maintained by fine regulation of iron absorption, circulation, utilization, and storage.[2,3] Primary (or genetic) HFE, caused due to mutation in HFE gene, leads to dysregulation of iron absorption through interactions with transferrin and hepcidin, leading to systemic manifestations of iron overload.[4,10] Hemostatic iron balance requires only 1–2 mg of iron per day; however, chronic disorders of erythropoiesis treated with iron and blood transfusions lead to significant iron accumulation.
As discussed earlier, liver, pancreas, heart, skin, and pituitary are some of the most common sites of iron deposition.[2-4] The presence of BBB does not allow deposition of excess iron in the CNS, but structures lying outside the BBB such as choroid plexus, pituitary, and circumventricular organs are commonly involved.[5-9] The hypogonadism is caused due to deposition of iron in anterior pituitary leading to malfunctioning of the gonadotrophs with relative sparing of the posterior pituitary seen.[1,10]
MRI has been an effective imaging modality for evaluating iron deposition. Excess iron leads to local distortions in the magnetic fields and spin relaxations, resulting in shortening of T2 relaxation time, leading to signal intensity similar to the flow void of adjacent internal carotid artery.[11] Brain MRI with a gradient response echo (GRE) sequence has shown heightened sensitivity in detecting CNS HFE. Susceptibility-weighted imaging (SWI) is a 3D-GRE MRI sequence with high spatial resolution and phase post-processing that improves the detection of paramagnetic properties, making it more sensitive for detecting hemosiderin compared to traditional 2D-GRE MRI.[12] Considering the artifacts caused by the air-filled sphenoid sinus encasing the sella turcica, SWI may not be ideal for assessing the pituitary gland. The decreased parenchymal enhancement of the anterior pituitary with sparing of posterior pituitary and stalk has shown consistency with hemosiderin deposition secondary to HFE.[13]
The deposition of iron within the anterior pituitary leads to signal intensity changes on T2 and T1W imaging. However, diagnosing pituitary gland lesions can be challenging given the diversity of lesions involving the pituitary gland [Table 1].[4,13] HFE is the only clinical entity associated with decreased signal intensity on T2W images and raised serum ferritin levels (usually >1000 ng/mL).[4]
| Differential diagnosis | Differentiating features |
|---|---|
| Pituitary hemochromatosis | History; biochemical correlation; decreased parenchymal enhancement with sparing of posterior pituitary and stalk. |
| Aneurysm | Continuous with internal carotid artery |
| Rathke’s cleft cyst or other mass lesion (Craniopharyngeoma or calcification or hemorrhagic pituitary adenoma) | Focal lesion; abnormal pituitary contour; mass effect on adjacent structures |
| Melanoma | History |
CONCLUSION
Considering diverse variety of lesions affecting the pituitary gland, adequate know how of the imaging manifestations along with clinical correlation is required to diagnose iron deposition in the pituitary gland. Hence, Contrast enhanced MRI should be used as primary non invasive tool in patients suspected of pituitary iron overload in setting of hypogonadotrophic hypogonadism.
TEACHING POINTS
The deposition of iron within the anterior pituitary leads to local distortions in the magnetic fields and spin relaxations, resulting in shortening of T2 relaxation time.
HFE is the only clinical entity associated with decreased signal intensity on T2W images and raised serum ferritin levels (Usually >1000 ng/mL).
The decreased parenchymal enhancement of the anterior pituitary with sparing of posterior pituitary and stalk has shown consistency with hemosiderin deposition, suggesting use of contrast-enhanced MRI as primary non-invasive tool in patients suspected of pituitary iron overload in setting of hypogonadotropic hypogonadism.
MCQs
-
Which of the following can cause pituitary HFE?
HFE gene mutation
Blood transfusions
Both of the above
None of the above
Answer Key: c
-
Which of the following is not an imaging finding of pituitary HFE?
Non-enhancement of pituitary stalk
Normal enhancement of posterior pituitary
Decreased T2 signal in anterior pituitary
Blooming in anterior pituitary
Answer Key: a
-
Which of the following do not cause decreased T2 signal intensity in the suprasellar region?
HFE
Hemorrhagic pituitary adenoma
Thrombosed internal carotid artery aneurysm
Melanoma
Answer Key: c
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- Hemochromatosis of the pituitary gland: MR imaging. Radiology. 1988;168:213-4.
- [CrossRef] [PubMed] [Google Scholar]
- Choroid plexus and pituitary gland hemochromatosis induced by transfusional iron overload: Two case reports. Radiol Case Rep. 2024;19:1666-70.
- [CrossRef] [PubMed] [Google Scholar]
- Case 179: Hereditary hemochromatosis. Radiology. 2012;262:1037-41.
- [CrossRef] [PubMed] [Google Scholar]
- Uptake and distribution of iron and transferrin in the adult rat brain. J Neurochem. 1992;59:300-6.
- [CrossRef] [PubMed] [Google Scholar]
- Brain iron homeostasis, the choroid plexus, and localization of iron transport proteins. Metab Brain Dis. 2009;24:673-84.
- [CrossRef] [PubMed] [Google Scholar]
- Transfusional hemochromatosis of the choroid plexus in beta-thalassemia major. J Comput Assist Tomogr. 2001;25:487-8.
- [CrossRef] [PubMed] [Google Scholar]
- The dark pituitary: Hemochromatosis as a lesser-known cause of pituitary dysfunction. J Belg Soc Radiol. 2022;106:63.
- [CrossRef] [PubMed] [Google Scholar]
- Transfusional iron overload and choroid plexus hemosiderosis in a pediatric patient: brain magnetic resonance imaging findings. Investig Magn Reson Imaging. 2019;23:390-4.
- [CrossRef] [Google Scholar]
- Hereditary hemochromatosis: Pathogenesis, diagnosis, and treatment. Gastroenterology. 2010;139:393-408. 408.e1-2
- [CrossRef] [PubMed] [Google Scholar]
- MR imaging findings of iron overload. Radiographics. 2009;29:1575-89.
- [CrossRef] [PubMed] [Google Scholar]
- Susceptibility-weighted MR imaging: A review of clinical applications in children. AJNR Am J Neuroradiol. 2008;29:9-17.
- [CrossRef] [PubMed] [Google Scholar]







